
Dosimeter badge services for medical, dental, and veterinary businesses
Learn how Radiation Detection Company’s easy-to-use dosimetry solutions can boost the efficiency of your practice.

Reviewed by
Chuck Nagel
Healthcare Business Partner
Last Updated: September 20, 2024
This post breaks down the key updates to the Mammography Quality Standards Act (MQSA) that went into effect on September 10, 2024, following the FDA’s final rule. It reviews how these changes impact radiation safety programs and dosimetry requirements throughout mammography departments, highlighting what interpreting physicians, technologists, medical physicists, and mammography facilities need to know to stay compliant, maintain high-quality imaging, and remain audit ready. For a more recent update on modern mammography workflows, read this blog post: How DBT, CEM, & Emerging Technologies Are Reshaping Mammography Radiation Safety.
The Mammography Quality Standards Act (MQSA) has served as the backbone of breast imaging regulation in the United States for over three decades. The 2024 amendment marks the first major overhaul since the law’s passage in 1992.
With recent updates, the FDA has modernized mammography oversight to reflect today’s technologies, patient expectations, and quality benchmarks. These updates introduce new federal requirements for breast density notification, reporting timelines, audits, and recordkeeping, marking a pivotal step toward greater transparency and consistency across all facilities nationwide.
The Mammography Quality Standards Act (MQSA) was enacted by Congress in 1992 to establish consistent, enforceable quality standards for mammography facilities across the United States. Its purpose was straightforward but critical: to ensure that all women, regardless of where they receive care, have access to safe, reliable, and high-quality mammography for the early detection of breast cancer.
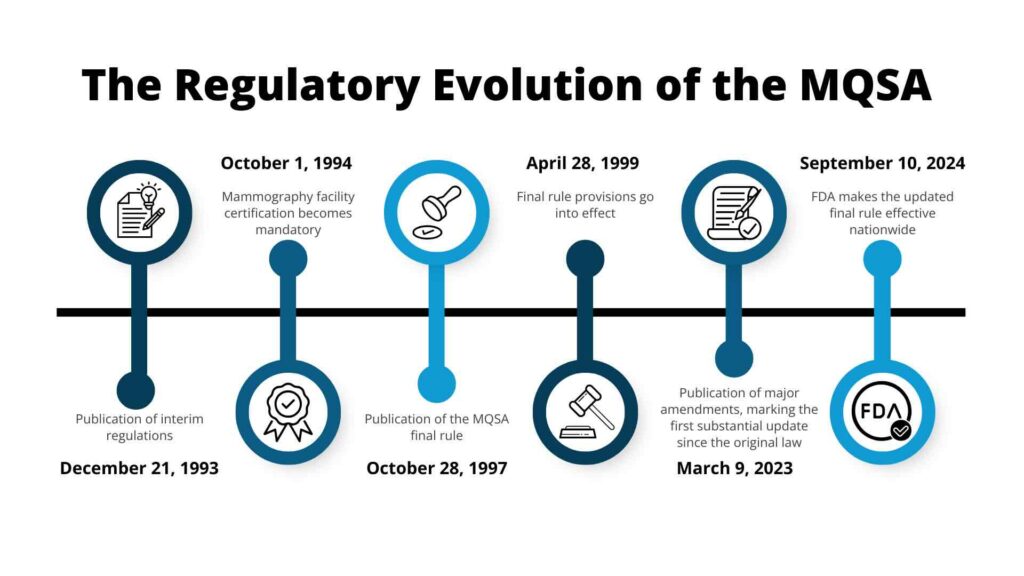
Key regulatory milestones include:
Together, these milestones illustrate how MQSA has evolved into the foundational regulatory framework that continues to shape and elevate the quality, safety, and consistency of mammography services throughout the country.
Mammography is a specialized form of breast imaging that uses low-energy X-rays to detect abnormalities long before they become clinically noticeable. While that definition hasn’t changed, what has changed is the regulatory framework governing how mammography is performed, interpreted, documented, and communicated.
Under the Mammography Quality Standards Act (MQSA), mammography is not simply an imaging exam – it is a federally regulated service with strict requirements designed to ensure patient safety, image quality, and diagnostic reliability. The 2024 MQSA amendments reinforce this shift by tying the clinical purpose of mammography (early, accurate detection of breast cancer) to updated expectations for reporting clarity, breast density assessment, outcomes auditing, and radiation safety practices.
In other words, mammography today is as much about the quality of the imaging ecosystem (equipment performance, technologist practices, interpreting physician accuracy, and recordkeeping integrity) as it is about the image itself.
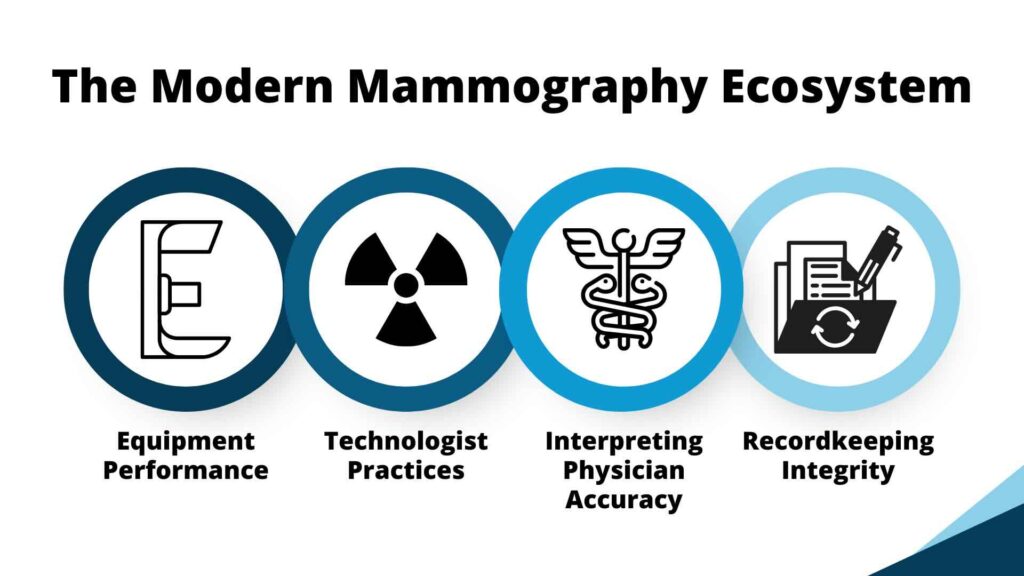
The MQSA final rule modernizes requirements, ensuring every facility meets the same national standards for transparency, consistency, and patient communication.
Since the MQSA was first issued in 1992, the field of mammography has undergone transformative change. Advances in imaging technology, such as digital mammography and digital breast tomosynthesis (DBT), have reshaped clinical practice, while growing patient awareness around breast density has driven demand for clearer, more consistent communication. At the same time, variation in state-level requirements and outdated federal processes highlighted the need for modernization.
The FDA’s updated final rule was designed to address these gaps by:
Together, these objectives set the stage for a more rigorous and consistent national framework – one that the 2024 MQSA amendment now formalizes through a series of new, clearly defined requirements.
The 2024 MQSA amendment introduced several new and enhanced requirements, effective September 10, 2024, designed to strengthen reporting clarity, facility oversight, recordkeeping practices, and overall quality assurance across mammography services.

Facilities must now meet expanded reporting and communication requirements, including:
These updates ensure patients receive timely, understandable results and consistent information across all facilities.
As of September 10, 2024, every facility must conduct an annual Medical Outcomes Audit capturing the following key performance metrics:
The audit must include all mammograms (screening and diagnostic) performed on or after the effective date, and facilities must retain MOA data until at least the next MQSA annual inspection.
Facilities must strengthen documentation practices and ensure continuity of patient access:
In other words, if the patient returns for additional mammograms within the 5- to 10-year window, the facility is required to keep the earlier exams only for a minimum of 5 years, because newer mammograms are on file and remain part of the permanent record.

If a facility closes or stops offering mammography services, it must:
Together, these requirements ensure that mammography records remain complete, traceable, and accessible throughout the patient’s care continuum, regardless of how long they remain with a facility or whether the facility continues operating.
The amendment expands the authority of FDA-approved accrediting bodies and state certification agencies. Their enhanced oversight includes:
This stronger enforcement framework underscores the FDA’s emphasis on consistent nationwide compliance.
While many MQSA personnel qualifications, equipment standards, and QA procedures remain intact, the updated final rule reinforces expectations for:
In practice, the most notable operational shifts relate to enhanced reporting, expanded audit requirements, and more stringent recordkeeping obligations.
Radiation safety regulations can be daunting to navigate. Our compliance experts built a Radiation Compliance Cheat Sheet to help you quickly identify and address gaps that can put your organization and employees at risk.
While the MQSA updates are regulatory in nature, their impact extends far beyond compliance.
The final rule strengthens the overall quality ecosystem of mammography by:
Collectively, these changes help ensure safer, higher-quality, and more transparent mammography services nationwide. Be sure to review the update summary from the FDA and consult the 2023 MQSA Final Rule in its entirety for more information.
The updated MQSA rule requires facilities to take a more proactive and structured approach to quality, documentation, and communication. To support readiness and long-term compliance, facilities should focus on the following strategic actions.

Review your current mammography program against all updated MQSA requirements, including reporting workflows, lay summaries, breast density notifications, MOA metrics, record retention, patient access processes, and staff training needs.
Revise reporting templates and lay summaries, adjust IT prompts for required elements, and update workflows for “Incomplete” assessments and MOA data capture.
Ensure interpreting physicians, technologists, QA staff, administrative/reporting teams, and leadership fully understand the new expectations, timelines, and documentation standards.
Establish or refine a process for collecting outcome data (PPV, CDR, recall rate), linking to pathology, tracking metrics annually, and reviewing results with leadership.
Confirm your retention timelines reflect federal and state requirements. Strengthen workflows for patient access, external record transfers, and facility closure/transition scenarios.
Use an updated MQSA compliance checklist to audit your policies, logs, audits, corrective action records, and MOA reports. Organize materials into a centralized compliance binder or digital folder.
Be proactive in communicating updates to your accreditation body and state certification agency to ensure alignment and satisfy any additional local requirements.
Schedule quarterly or semiannual reviews of MOA metrics, implement corrective actions where needed, and document improvements thoroughly.
Review all patient-facing materials – including letters, summaries, website content, consent forms – to confirm clarity, readability, and accuracy of breast density and exam information.
Monitor evolving FDA guidance, accrediting body updates, and any applicable state law changes to keep your compliance program current.
With these operational steps in place, facilities are better positioned not only to meet the updated MQSA requirements but also to understand how these changes ripple into broader areas of radiation safety, personnel monitoring, and dosimetry practices – topics we’ll explore more in the following section.
Although the 2024 MQSA amendment centers on image quality, reporting, and patient communication, its operational implications extend deeply into radiation safety infrastructure. The new requirements increase the burden of documentation, elevate expectations for personnel monitoring, and reinforce the need for integrated, data-driven dosimetry systems. The following areas represent the most significant impacts on radiation protection programs across mammography facilities.
The updated MQSA framework places a strong emphasis on verifiable, auditable records. Every component of mammography operations, including radiation exposure, equipment performance, and physicist evaluations, must now be traceable and readily available for FDA or accrediting body review.
Facilities must maintain:
These expectations significantly reduce the feasibility of paper-based logs or decentralized recordkeeping. As a result, facilities are moving toward digital dosimetry dashboards, automated report generation, and secure cloud-based storage that ensures data integrity and rapid retrieval during inspections.
See how RDC simplifies compliance through MyRadCare™ – our secure customer platform offering 24/7 access to dose reports, badge management, and audit-ready documentation.
Under strengthened oversight, inspectors will focus more closely on the adequacy and rigor of radiation protection programs.

Mammography facilities should expect greater scrutiny of:
These elements are increasingly recognized as essential to establishing a defensible compliance posture. Organizations can proactively meet these requirements by using NVLAP-accredited dosimeters that deliver exceptional performance across the 16 (32 kVp) to 40 keV (80 kVp) range.
The new Medical Outcomes Audit (MOA) requirement introduces a more data-centric model of quality oversight. While MOA focuses on diagnostic performance, it has indirect implications for radiation monitoring: facilities must ensure that their radiation safety data can withstand the same level of scrutiny as their clinical performance metrics.
To meet these expectations, facilities are seeking:
For a deeper dive into building a compliant, efficient radiation monitoring program, explore our Complete Dosimetry Program Management Guide.
While MQSA focuses primarily on patient imaging standards, the rule indirectly heightens expectations around staff exposure control. Mammography technologists – particularly those performing digital breast tomosynthesis (DBT) or working in high-volume centers – may face increased scatter radiation compared to historical film systems.

This reinforces the need for:
The MQSA amendments elevate radiation monitoring from a background administrative task to a frontline compliance deliverable.
The updated MQSA framework represents one of the most substantial shifts in mammography regulation in more than two decades. While the foundational requirements for personnel, equipment, and image quality remain intact, the expectations around reporting, patient communication, record retention, outcomes auditing, and accreditation oversight have expanded dramatically. These updates reflect a broader move toward transparency, standardization, and data-driven oversight across mammography departments.
For facilities, the opportunity lies in approaching these changes not as a regulatory burden, but as a catalyst for operational improvement. By strengthening documentation practices, modernizing workflows, and enhancing radiation safety programs, facilities can reduce regulatory risk, demonstrate continuous quality improvement, and reinforce their commitment to high-quality breast imaging.
Radiation Detection Company supports this mission by ensuring seamless, audit-ready dosimetry program management. With reliable badge delivery, industry-leading customer service, and 24/7 access to digital dose reports through our MyRadCare™ platform, we help facilities maintain uninterrupted compliance and keep their radiation protection programs inspection-ready at all times.
To learn how RDC can support your facility through the new MQSA landscape, contact Radiation Detection Company or call 800.250.3314.
The 2024 MQSA amendment is the first major update to mammography regulation in more than 25 years. The rule introduces new federal requirements for breast density notification, faster reporting timelines, and annual Medical Outcomes Audits (MOA) tracking key performance metrics (PPV, CDR, recall rates). It also strengthens expectations for recordkeeping, patient access, and accreditation oversight, bringing national standards in line with modern imaging technologies and today’s patient communication expectations.
The updated rule places greater scrutiny on documented radiation protection programs. Facilities must maintain:
These records must be easily accessible during FDA or accrediting body inspections. In response, many facilities are adopting digital dosimetry dashboards, automated reporting tools, and cloud-based recordkeeping to ensure full traceability and rapid retrieval of radiation monitoring data.
At Radiation Detection Company, we offer centralized, exception-based reporting through MyRadCare Insights™ – a powerful radiation safety analytics platform designed for healthcare systems that demand better visibility, faster decision-making, and lower compliance risk.
Under MQSA’s enhanced audit and documentation requirements, facilities benefit from radiation monitoring systems that provide:
Integrated digital dosimetry systems help facilities streamline documentation, reduce administrative burden, and demonstrate compliance during inspections.
If a facility cannot demonstrate compliance with reporting, recordkeeping, QA, or audit requirements, inspectors may:
The most effective protection is proactive preparation: routine internal audits, comprehensive staff training, updated documentation workflows, and verified QA records. These steps help ensure continuous compliance and uninterrupted mammography services.

Learn how Radiation Detection Company’s easy-to-use dosimetry solutions can boost the efficiency of your practice.